CATEGORIES
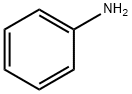
Aniline
English Name: Aniline
MF: C6H7N
MW: 93.13
CAS: 62-53-3
Aniline is the simplest primary aromatic amine, a compound formed by replacing one hydrogen atom in the benzene molecule with an amino group. It is a colorless, oily, flammable liquid with a strong odor. Its melting point is -6.3℃, boiling point is 184℃, relative density is 1.0217 (20/4℃), refractive index is 1.5863, flash point (open cup) is 70℃, autoignition point is 770℃, and it decomposes at 370℃. It is slightly soluble in water and readily soluble in organic solvents such as ethanol, ether, and chloroform. It turns brownish-white upon exposure to air or sunlight. It can be steam distilled, with a small amount of zinc powder added during distillation to prevent oxidation. Purified aniline can be treated with 10–15 ppm of NaBH4 to prevent oxidation and deterioration. Aniline solutions are alkaline and readily form salts with acids. The hydrogen atom on its amino group can be replaced by a hydrocarbon or acyl group to form secondary or tertiary anilines and acylanilines. Substitution reactions primarily produce ortho- and para-substituted products. It reacts with nitrous acid to form diazonium salts, from which a series of benzene derivatives and azo compounds can be prepared.

Aniline is an important chemical raw material, with over 300 major products derived from it. It is mainly used in MDI, the dye industry, pharmaceuticals, and as a rubber vulcanization accelerator, such as p-aminobenzenesulfonic acid in the dye industry and N-acetanilide in the pharmaceutical industry. It is also a raw material for manufacturing resins and coatings. In 2008, aniline consumption was approximately 360,000 tons, and the estimated demand for 2012 was approximately 870,000 tons, while the commercial aniline production capacity reached 1.37 million tons, resulting in an overcapacity of nearly 500,000 tons. Aniline is highly toxic to blood and nerves, and can cause poisoning through skin absorption or inhalation. Industrially, aniline is mainly produced using two methods: 1. Hydrogenation of nitrobenzene via active copper catalysis; this method allows for continuous production and is pollution-free. 2. Reaction of chlorobenzene and ammonia at high temperature in the presence of a copper oxide catalyst.
Uses
Aniline is one of the most important intermediates in the dye industry, and a major raw material for pharmaceuticals, rubber accelerators, and antioxidants. It can also be used to make fragrances, varnishes, and explosives. Aniline is used in the manufacture of dyes, pharmaceuticals, resins, varnishes, perfumes, vulcanized rubber, and even solvents. It is a hazardous and harmful substance affecting the early life stages of marine animals. According to the U.S. Environmental Protection Agency (EPA), it is a candidate compound for environmental and food contaminants, and a contaminant for drinking water (Chemicalbook 3).
Aniline is an important raw material for pesticide production. From aniline, N-alkylaniline, alkylaniline, o-nitroaniline, o-phenylenediamine, phenylhydrazine, cyclohexylamine, etc., can be derived. It can be used as an intermediate in the production of fungicides such as sodium chlorothalonil, seed dressing agent, methyl thiophanate, thiophanate-methyl, carbendazim, pyraclostrobin, and benomyl; insecticides such as triazophos, pyridazin, and quinalphos; and herbicides such as metolachlor, acetochlor, butachlor, cycloazinone, and imidacloprid.
CONTACT US
Please use the form below to get in touch.
If you need a reply we will get in touch as soon as possible.