CATEGORIES
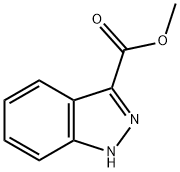
1H-indazole-3-carboxylic acid methyl ester
English Name: 1H-indazole-3-carboxylic acid methyl ester
Chinese Name: 1H-indazole-3-carboxylic acid methyl ester
MF: C9H8N2O2
MW: 176.17
CAS: 43120-28-1
Production Method
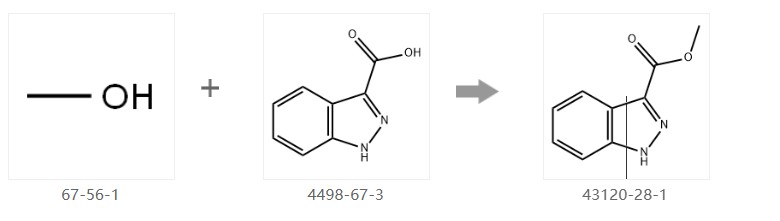
Indazole-3-carboxylic acid (5.0 g, 30.8 mmol) was dissolved in methanol (50 mL) at 0 °C, and thionyl chloride (15 mL) was slowly added dropwise. After the addition was complete, the reaction mixture was heated to reflux temperature and maintained at this temperature for 1.5 hours. After the reaction was complete, the reaction mixture was concentrated under reduced pressure to obtain a crude product. The crude product was neutralized with saturated sodium bicarbonate solution (50 mL), followed by extraction with ethyl acetate (50 mL × 3). The combined organic phases were dried with anhydrous sodium sulfate. The drying agent was removed by filtration, and the filtrate was concentrated under reduced pressure to give methyl 1H-indazole-3-carboxylic acid as a white solid (5.1 g, 94% yield). The proton NMR spectrum (300 MHz, d6-DMSO) data are as follows: δ 13.91 (s, 1H), 8.06 (d, J = 8.2 Hz, 1H), 7.65 (d, J = 8.4 Hz, 1H), 7.44 (ddd, J = 8.3 Hz, 6.9 Hz, 1.1 Hz, 1H), 7.30 (dd, J = 7.9 Hz, 6.9 Hz, 0.9 Hz, 1H), 3.92 (s, 3H).
CONTACT US
Please use the form below to get in touch.
If you need a reply we will get in touch as soon as possible.